题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
已知Mg(s)→Mg(g) ΔH1=147kJ·mol-1 Mg(g)→Mg2+(g)+2e- ΔH2-2188kJ·mol-1 S(s)→S(g) △H3=277kJ·mol-1 S
已知Mg(s)→Mg(g) ΔH1=147kJ·mol-1
Mg(g)→Mg2+(g)+2e-ΔH2-2188kJ·mol-1
S(s)→S(g) △H3=277kJ·mol-1
S(g)+2e-→S2-(g) △H4=332kJ·mol-1
Mg(s)+S(s)→MgS(s) ΔH5=-347kJ·mol-1
计算MgS的晶格能U。
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案


 更多“已知Mg(s)→Mg(g) ΔH1=147kJ·mol-1 …”相关的问题
更多“已知Mg(s)→Mg(g) ΔH1=147kJ·mol-1 …”相关的问题
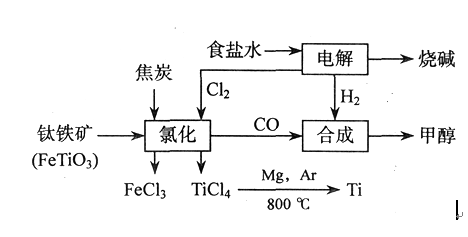 请填写以下空白:
请填写以下空白: