 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
Fire engines and ambulances have ______ over othertraffic.A) priorityB) urgencyC) supe
Fire engines and ambulances have ______ over othertraffic.
A) priority
B) urgency
C) superiority
D) emergency
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案
 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
Fire engines and ambulances have ______ over othertraffic.
A) priority
B) urgency
C) superiority
D) emergency
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案
 更多“Fire engines and ambulances ha…”相关的问题
更多“Fire engines and ambulances ha…”相关的问题
Which of the following statements is NOT true according to the passage?
A.Color probably has an effect on us which we are not conscious of.
B.Yellow fire engines have caused many bad accidents in some advanced communities.
C.People exposed to pure blue start to breathe more slowly.
D.The psychology of color is of some practical use.
A typical combustion process is the burning of gasoline in an automobile engine. The vaporized fuel is mixed with air, compressed in the engine's cylinder, and ignited by a spark. As the fuel flame up, the heat produced flows into the adjacent layer of unburned fuel and ignites it. In this way a zone of fire spreads throughout the fuel mixture is called a combustion wave.
The speed at which such a combustion wave travels through a fuel mixture is called the burning velocity of the mixture. The burning velocity of a gas such as methane quietly burning in air is only about one foot per second. By comparison, the burning velocity of more reactive combinations such as the rocket Fuels, hydrogen and fluorine, can be hundreds of feet per second.
If the fuel flows at the same speed as the combustion wave, the result is a stationary flame, like the one in your kitchen gas burner. In the kitchen burner a jet of gas mixed with airflows from the opening in the head of the burner. If the velocity of the fuel mixture flowing from the opening is greater than its burning velocity, the flame blows out.
In jet engines speeding through the air at 500 to 600 miles per hour, the engine's flame is sometimes blown out by the blast of air entering the combustion chamber at high speeds. Jet pilots call this condition "flameout".
Combustion can sometimes occur very slowly. A familiar example of slow combustion is the drying of ordinary oil-based paint. In this chemical reaction, called oxidation, the oxygen in the air reacts with the drying oil in the paint to provide a tough film. The linseed oil molecules link together, forming an insoluble coating.
How can the chemical reaction involved in such a quiet process as the drying of paint also produce spectacular flames and explosions? The main difference between the two is the temperature at which they occur.
At lower temperatures the reaction must take place over a long time. The heat which is slowly produced is dissipated to the surroundings and does not speed up the reaction. When the heat produced by the low-temperature reaction is retained instead of being dissipated, the system breaks into flame.
In a flame or explosion, the reactions are extremely fast. In many chemical processes, however, such a rapid oxidation process would be extremely destructive.
Which of the following states the major difference between oxidation and fire?
A.Their burning velocities differ in rate.
B.Oxidation is a chemical reaction while fire is a physical reaction.
C.Oxidation does not create heat.
D.They occur at different temperatures.
A.see
B.reckon
C.distinguish
D.sense
According to the author, a born naturalist should first of all be ______. ()
A.full of ambition
B.knowledgeable
C.full of enthusiasm
D.self-disciplined
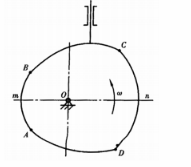
图(a)所示为一偏置直动推杆盘形凸轮机构,凸轮轮廓上的AmB和cnD为两段圆心位于凸轮回转中心的圆弧,凸轮转向如图所示。试在图上标出推程运动角δ0、回程运动角δ'0、推杆升程h以及推杆与凸轮在c点接触时的压力角α。
计算曲线积分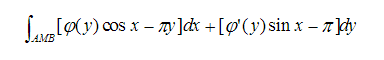 ,其中AMB为连接A(π,2)与点B(3π,4)的线段
,其中AMB为连接A(π,2)与点B(3π,4)的线段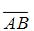 之下方的任意分段光滑简单闭曲线,且该路线与线段
之下方的任意分段光滑简单闭曲线,且该路线与线段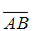 所围图形面积为2。
所围图形面积为2。